|
All three orbitals need to be drawn even if one or more. Now it is possible to find the orbital notation of silver very easily through electron configuration. Melting point The temperature at which the. carbon atoms and single bonds to the hydrogen atoms. 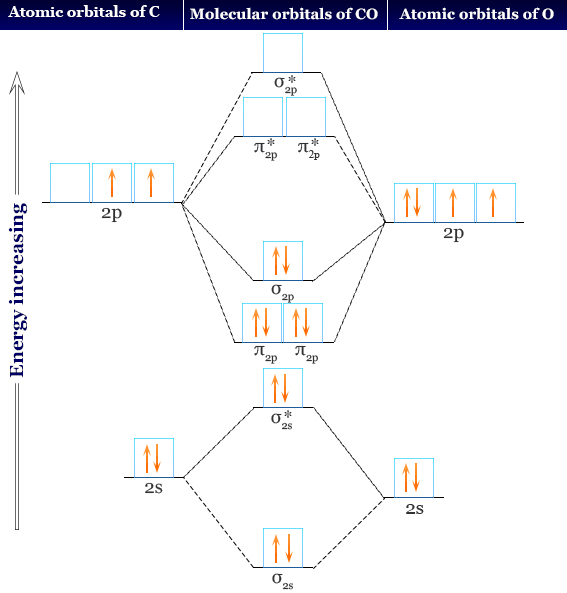
Electron configuration The arrangements of electrons above the last (closed shell) noble gas. Elements are organised into blocks by the orbital type in which the outer electrons are found. Use the orbital diagrams already done to write the electron configuration for. 
Based on Hund's rule, one electron fills each p \rm p p orbital, and each electron has the same spin. Element Carbon (C), Group 14, Atomic Number 6, p-block, Mass 12.011. electron configuration notation for carbon is 1s2 2s2 2p2. Using arrows to show the spin orientation of each electron, the orbital diagram is often shown this way: The single electrons in the two p-orbitals is in accordance with Hunds Rule. Write the electron configuration for each of the following ions: As 3+. an orbital diagram or an electron configuration 11. In the explanation below, I show a common means of diagramming this. Draw the orbital energy diagram, for Na and S-2, putting the valence. These three electrons have unpaired spins. 
The electron configuration and orbital diagram for carbon are: Nitrogen (atomic number 7) fills the 1s and 2s subshells and has one electron in each of the three 2p orbitals, in accordance with Hund’s rule. Four of them fill the 1s and 2s orbitals. 
Following the 2s sublevel is the 2p, and p sublevels always consist of three orbitals. For example, the ground state electron configuration of nitrogen (1 s 2 2 s 2 2 p 3 \rm 1s22s22p3 1 s 2 2 s 2 2 p 3) indicates that it has 3 3 3 electrons occupying the 2 p 2 \rm p 2 p orbital. If we added one more electron to borons orbital diagram we will get carbons orbital diagram. Carbon (atomic number 6) has six electrons. However, the diagram above clearly shows that the 4s orbital is filled before the 3d orbital. And, as we stated above in the Aufbau principle, electrons fill lower energy orbitals before filling higher energy orbitals. Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. Represent the organization of electrons by an electron configuration and orbital diagram. Figure : The ground state electron configuration of carbon, which has a total of six electrons. The lowest energy sublevel is always the \(1s\) sublevel, which consists of one orbital.\)). \) shows the order of increasing energy of the sublevels.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
